Introduction
Food-contact sealing gaskets are often underestimated in system design, yet their compliance level directly determines whether a product can enter food, beverage, and pharmaceutical supply chains. For procurement teams, the real risk is not price, but whether materials, processes, and documentation can withstand FDA audits and traceability checks.Based on long-term cooperation with food equipment manufacturers, Xiamen Ruicheng has found that most food-grade gasket failures stem from “seemingly compliant” injection molding solutions that lack a complete evidence chain.
In injection molded parts manufacturing, isolated parameters are no longer sufficient to support purchasing decisions; what truly matters is an end-to-end solution covering material selection, manufacturing control, validation, and delivery.Building trust from the compliance source is the core logic of food-grade sealing gasket injection molding.
How to Select Injection Molding Materials That Meet FDA Requirements for Sealing Gaskets?
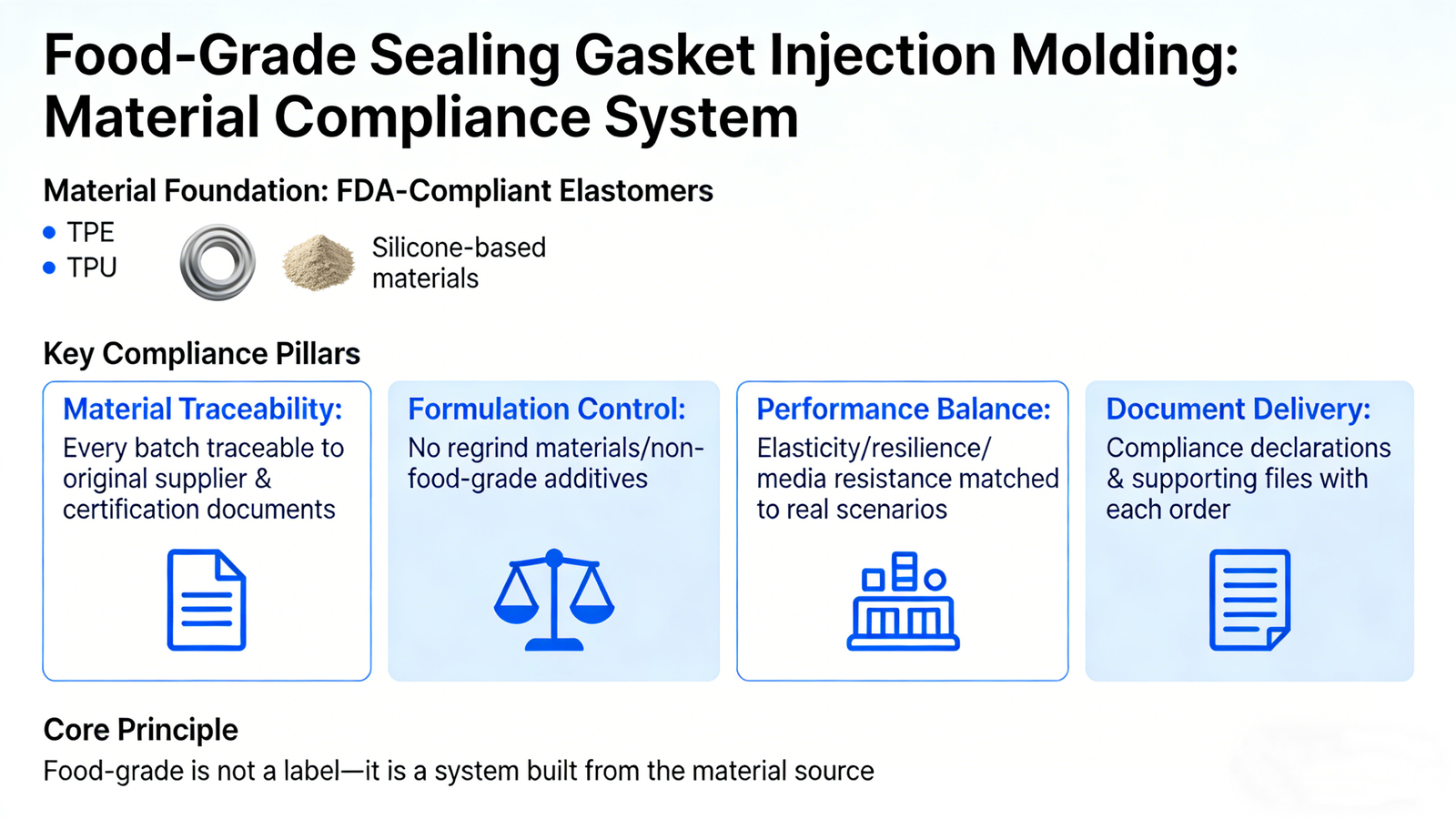
In food-grade sealing gasket injection molding, the material defines the upper limit of compliance. Xiamen Ruicheng prioritizes FDA-compliant elastomers such as TPE, TPU, and silicone-based materials, all supported by original manufacturer MSDS files and food-contact declarations. Only when the base material itself meets FDA food-contact compliance does injection molding truly become meaningful. During project onboarding, we align material selection with FDA 21 CFR 177 clauses and evaluate suitable food-grade elastomers to eliminate substitution risks at the source.
Material traceability: Every batch is traceable to the original supplier and certification documents, ensuring audit continuity.
Formulation control: Regrind materials and non-food-grade additives are strictly prohibited to prevent migration risks.
Performance balance: Elasticity, resilience, and media resistance are matched to real application scenarios rather than isolated hardness values.
Document delivery: Compliance declarations and supporting files are delivered with each order to simplify customer audits.
😊 Food-grade is not a label—it is a system built from the material source.
How Can Injection Molding Processes Prevent Compliance Risks in Food-Grade Sealing Gaskets?

Even with compliant materials, improper injection molding processes can compromise food-grade properties. Xiamen Ruicheng strictly controls processing aids, demolding methods, and cross-contamination risks throughout injection molded parts manufacturing. The essence of process control is preventing any non-food-grade variables from entering the gasket surface or internal structure. In practice, we implement clean injection molding workflows and reference ISO 14644 cleanroom management concepts to create a stable and repeatable production environment.
Equipment segregation: Dedicated machines and mold management systems are used for food-grade projects to avoid cross-line contamination.
Process windows: Verified molding temperatures and pressure profiles reduce thermal degradation risks.
Surface control: Secondary coatings are minimized; surface quality is achieved through mold precision.
Process records: All critical parameters are documented to support quality traceability.
🧩 True food-grade compliance is reflected in every consistently repeated molding cycle.
How Can Injection Molded Sealing Gaskets Be Verified as Truly FDA Food-Grade?

For buyers, “FDA compliant” is not a verbal promise but a verifiable, deliverable evidence system. Xiamen Ruicheng completes internal and third-party validation loops before shipment. Only when test data and documentation are aligned does a food-grade sealing gasket become commercially viable. We integrate SGS testing and recognized food contact compliance processes to reduce audit burdens for customers.
Type testing: Migration and sensory tests are performed based on actual contact media, avoiding one-size-fits-all certificates.
Batch consistency: First article approval and in-process inspections ensure mass-production stability.
Documentation support: Assistance is provided for FDA inspections and end-brand audits.
Risk anticipation: Potential failure points are identified during pilot runs to minimize rework during scale-up.
🔍 Compliance is not a one-time test, but a sustainable validation system.
Key Compliance Factors Comparison for Food-Grade Sealing Gasket Injection Molding
| Dimension | Conventional Gaskets | Claimed Food-Grade | Systematic Food-Grade Solution | Xiamen Ruicheng Solution |
|---|---|---|---|---|
| Material Source | Not traceable | Single declaration | OEM-certified | Full traceability |
| Process Control | Standard | Partial limits | Clean management | Dedicated system |
| Testing Support | None | One-time | Application-based | Scenario-driven |
| Audit Support | None | Passive | Basic | Proactive |
If you are reassessing long-term compliance risks for food-grade sealing gaskets, now is the right time to revisit your sourcing strategy—contact us.
From Compliance to Mass Production: Decision-Support Value
Food-grade sealing gasket injection molding is not a one-off purchase but an ongoing partnership throughout the product lifecycle. Xiamen Ruicheng focuses on real decision pressures during selection, validation, scale-up, and repeat orders, reducing overall compliance and communication costs through early-stage engineering involvement. In injection molded parts manufacturing, we treat engineering support as part of the product itself.
1.Selection support: Early-stage material and structural evaluation during design.
2.Pilot validation: Small-batch trials replace high-risk direct mass production.
3.Documentation coordination: Support for internal and customer compliance documentation.
4.Continuous optimization: Process refinement based on real-world application feedback.
FAQ – Procurement-Oriented Questions
Question: What is the quality baseline for your food-grade sealing gasket injection molding?
Answer: We base our standards on FDA 21 CFR requirements, supported by OEM material certifications, controlled process records, and third-party test reports, ensuring a fully auditable compliance chain.
Question: What information is required to start sourcing food-grade sealing gaskets?
Answer: Application media, operating temperature, cleaning methods, and expected service life are recommended inputs, enabling fast material matching and quotation.
Question: How are lead times managed for different order volumes?
Answer: Small-batch pilot runs are supported for validation, while mass production benefits from stable capacity planning and delivery schedules.
Question: How are compliance or quality disputes handled after delivery?
Answer: A clear issue evaluation process is applied, followed by rapid corrective actions such as rework or replacement when responsibility is confirmed.
Question: Do you support customized food-grade solutions?
Answer: Yes, customized materials, tooling, and testing plans are developed based on specific operating conditions and certification requirements.
Conclusion
Food-grade sealing gasket injection molding is fundamentally a system built on trust and evidence. By integrating material control, process discipline, and validation mechanisms, Xiamen Ruicheng helps customers reduce compliance uncertainty. A truly reliable FDA food-grade solution should withstand scrutiny before it ever reaches the market. When injection molded parts manufacturing shifts from “parameter display” to “decision support,” long-term partnership value naturally emerges.
For expert assistance in implementing solutions for your production needs, visit our resource center or contact us. Let’s help you scale up your manufacturing with precision and efficiency!







